On Wednesday, Pfizer and partner BioNTech, makers of the United States’ first Food and Drug Administration (FDA) emergency authorized COVID vaccine, saw their share prices plummet as discouraging new research from the Mayo Clinic forced investors to question how long the Pfizer vaccine remains effective at preventing coronavirus infections and protecting those who are vaccinated from getting sick with a Delta variant case.
Pfizer’s shot may be significantly less effective than Moderna’s against breakthrough infections (42% efficacy for Pfizer/BioNTech versus 76% for Moderna), according to the data, and Pfizer stock ended the day down nearly 4% while Germany’s BioNTech slipped 13.76%. But the urgency of the investor reaction underscores the gravity of the Delta wave and uncertainty surrounding a pioneering vaccine that’s been on the U.S. market for exactly nine months.
One key issue raised by the study, which has yet to be published in a peer-reviewed medical journal, for the millions of Americans who received two doses of Pfizer’s treatment is whether or not a Pfizer booster dose—either of the currently available jab or a new and updated version—is necessary to keep up with mutations or prevent COVID-19 reinfection from older coronavirus strains. Given that Pfizer expects to have delivered 500 million doses of its COVID vaccine under supply agreements with the U.S. government by the spring of 2022, should Americans be worried about catching a nasty case of the Delta variant or spreading the pathogen to others if they’re immunized with Pfizer’s product?
In short: There is now mounting evidence that mRNA-based vaccines such as Pfizer’s and Moderna’s lose potency over time and especially against the Delta variant, and that the Pfizer vaccine’s efficacy drop is significantly more dramatic. The Mayo Clinic study noted a wide shortfall in the mRNA vaccines’ ability to prevent infections among patients using the Mayo Clinic Health System for the month of July, when Delta variant cases made up more than 70% of new local infections in its home state of Minnesota, compared with earlier in the year. Between January and July, Moderna’s jab was 86% effective at preventing infection in this population while Pfizer’s was 76% effective. But for July alone, those numbers fell to 76% for Moderna and 42% for Pfizer, and the researchers observed similar drops for the Pfizer shot outside of Minnesota in states with high COVID counts such as Florida. (On a brighter note, both the Moderna and Pfizer vaccines still proved highly effective at preventing the need for COVID infection-related hospitalization at 92% and 85% efficacy, respectively.)
Those are concerning figures. But it’s important to note several caveats. For one, we still need more data on how the Delta variant is shaping the pandemic and interacting with available COVID treatments given that mutation’s surge is still relatively new. Collecting this information has a long lag time in the U.S. as local information trickles up to the Centers for Disease Control (CDC) database, and the agency’s decision in May to stop tracking breakthrough infections that may cause symptoms but not coronavirus-related hospitalization or death among the fully vaccinated has created an information black hole. Furthermore, tracking how strong a vaccine is against any viral strain over an extended period, and how much immunizing power it may lose and how quickly, is inherently handicapped by having to monitor how the situation plays out on the ground in real time.
But public health experts note that if this trend continues to hold true in more peer-reviewed research, drugmakers and medical institutions will have to rethink their approach to fighting the highly infectious Delta variant, potentially through boosters, modified vaccines specifically tailored toward strains like Delta, or by trying different combinations of COVID vaccines as the U.K. has done with AstraZeneca’s shot in concert with an mRNA-based vaccine.
Prominent physician-scientist and Scripps Research Translational Institute founder and director Eric Topol didn’t mince words about the steady drip of new information on mRNA jabs such as Pfizer’s and how they fare against this vicious variant.
“There needs to be truth-telling about the reduced protection of mRNA vaccines vs symptomatic Delta infections. It was 95% pre-Delta. Many are claiming it’s still ~80%. It isn’t. 50-60% is best estimate from all sources (not US, since we don’t have the data),” wrote Topol in a tweet on Wednesday, attaching a table he compiled of the available efficacy data on mRNA vaccines and the Johnson & Johnson and AstraZeneca jabs when it comes to fighting the Delta strain.
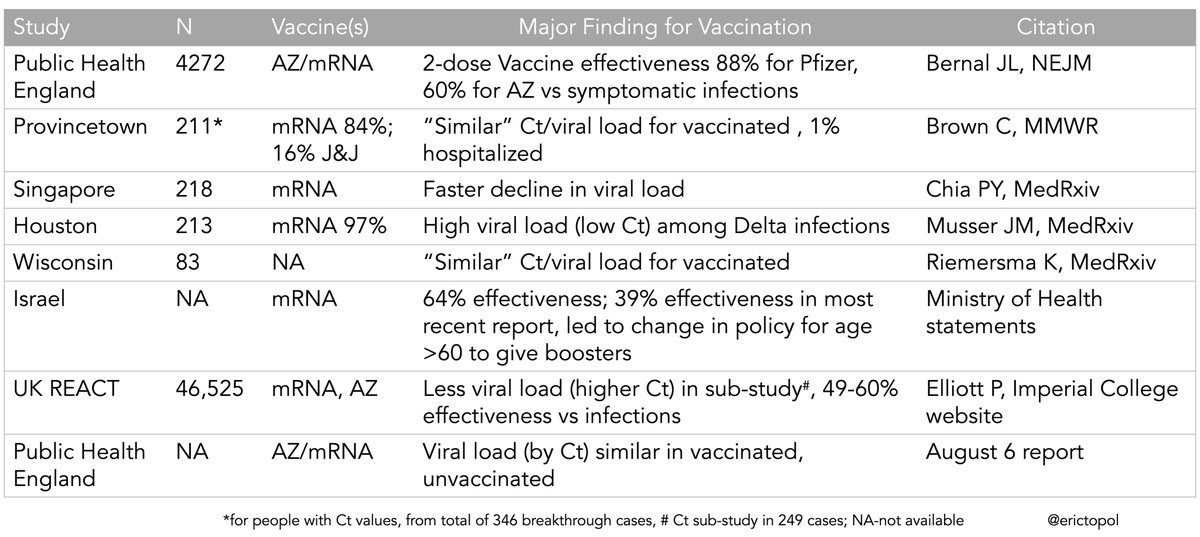
Topol goes on to say he expects the numbers in the U.S., once more of them come out, to be even worse given the experiences of more vaccinated countries like Israel, which are keeping close tabs on how strong Pfizer’s mRNA immunization remains over time. While the prevention of hospitalization and death is still an undeniable win, the prospect of getting sick for a few days or weeks is still cumbersome and a public health concern for high-risk individuals such as the immunocompromised.
The overarching message for those worried about their Pfizer vaccine being ineffective against the Delta variant is to practice public safety guidelines such as wearing masks around others indoors and maintaining distance in crowded areas, as well as commonsense practices like covering your mouth if you cough or sneeze and keeping stuffy, crowded rooms well ventilated on top of masking. And if you haven’t been vaccinated yet, there’s no time like the present. The three COVID shots from Pfizer, Moderna, and Johnson & Johnson are all available to most of the public (including for those 12 years and older for the Pfizer vaccine), and getting fully immunized now would buy precious time as drugmakers and government agencies figure out the best way to proceed with boosters.
On that note, Pfizer and Moderna have stated that their COVID vaccines will likely require booster doses down the line, possibly before winter 2021, and especially for people who are four, six, or nine months out from when they became fully vaccinated or have high risk for serious COVID illness. The companies are conducting booster trials of both their existing vaccines and new types, with Moderna reporting “robust” immunization from three separate, experimental booster candidates among those who had already been vaccinated. Pfizer and BioNTech have touted increased antibody response against other coronavirus variants after a third dose of their existing vaccine and believe they will see similar effects against Delta. Pfizer is expected to request FDA authorization for a third dose this month and has already applied for full approval of its vaccine.
Furthermore, Pfizer/BioNTech said in a statement they expect to be able to “develop and produce a tailor-made vaccine against that [Delta] variant in approximately 100 days after a decision to do so, subject to regulatory approval” given the highly modifiable nature of mRNA-based treatments. But we’re still a ways from knowing whether the mix-and-match shots approach, the stay-the-course-on-current-jabs method, or a new kind of booster shot entirely will ultimately tame the COVID Delta variant.
Subscribe to Fortune Daily to get essential business stories straight to your inbox each morning.












