Happy Monday. Last Monday, I offered a dark scenario to start off your workweek: the prospect of a highly pathogenic zoonotic virus spreading feverishly around the globe—or, as I should have titled the column: “Hi, everyone. How was your weekend?”
Today, I am determined to keep things upbeat—so again, let’s discuss the ongoing threat of pandemic. But this time, here’s a far more hopeful spin: It may well be that the oldest threat to humankind—the spread of infection—is the one where the newest digital health technology can have the greatest impact.
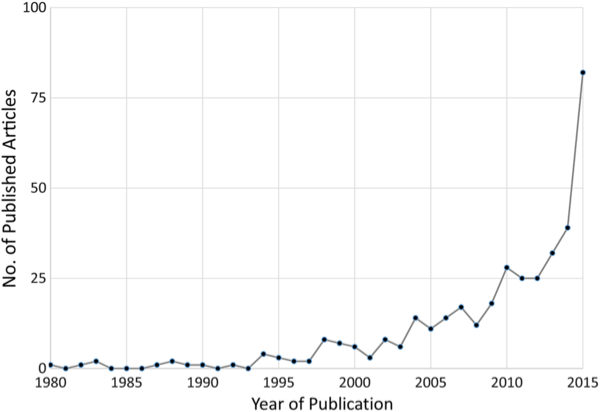
The Journal of Infectious Diseases, the flagship publication of the Infectious Diseases Society of America, devoted its December issue to ways in which big data—and a host of new electronic tracking platforms for both medical and non-medical events—can be used to vastly improve our traditional disease surveillance systems (which, frankly, are terribly flawed now). And with the number of scholarly articles on the subject having risen exponentially since 2001 (see the chart below), there’s a tremendous amount of information out there about what works and what doesn’t.
From: Big Data for Infectious Disease Surveillance and Modeling
As Jihye Choi and colleagues at Seoul National University’s School of Public Health reviewed in another journal this past December (really worth reading), there are three fundamental limitations of many current disease-surveillance systems. The first is language interoperability. Most tend to be in English—even when the centers of outbreaks are frequently in non-English-speaking regions. Second, the networks are often “excessively complex” requiring costly upkeep. And finally, these largely passive systems, run by national public health authorities, often lag in reporting local events—a big problem when speed is of the essence. (That’s what we saw, for example, in the latest Ebola outbreak in West Africa.)
Two new approaches worth checking out are ResistanceOpen, developed by a team of epidemiologists, clinicians, and data experts at Boston Children’s Hospital, and the Global Virome Project.
The first is a very cool interactive global map tracking reports of antimicrobial resistance, based on both public records and data submitted by independent users of this “open-collaboration database.” (Want a cheap thrill? Type in your zip code here and discover what superbugs are lurking in your backyard.)
The second is a more comprehensive ten-year project—proposed by a world-spanning alliance of experts—to preempt emerging infectious disease threats by characterizing virtually all zoonotic viruses that have epidemic potential—and creating a real-time, open-access database to monitor them. It’s an ambitious—and worthwhile—endeavor. (You can read more about it here.)
Sy has the day’s news below.
| Clifton Leaf | |
| @CliftonLeaf | |
| clifton.leaf@fortune.com |
DIGITAL HEALTH
Google's DeepMind aims to bring the (sort of) blockchain to health care. Add one more big name to the burgeoning list of companies wading deeper into the blockchain arena: Google's London-based AI arm DeepMind. DeepMind is already involved in health care projects with the U.K.'s National Health Service. In a blog post, DeepMind Co-Founder & Head of Applied AI Mustafa Suleyman and Head of Security and Transparency Ben Laurie explained what the new auditing project, which involves a digital ledger and tracking system similar to the blockchain tech at the heart of Bitcoin, will entail. "Like blockchain, the ledger will be append-only, so once a record of data use is added, it can’t later be erased. And like blockchain, the ledger will make it possible for third parties to verify that nobody has tampered with any of the entries," they wrote. The group believes it's important to ramp up these auditing and security technologies to both ensure confidence and provide real-time information about when and why a piece of data is used in the health system. (WIRED)
Get a genetic test - or your insurance costs go up. A bill which advanced through a House committee last week could allow employers to put intense pressure on their workers to take a genetic test - and those who refuse would have to pay significantly more for their company-provided health care. The so-called Preserving Employee Wellness Programs Act would nix certain privacy protections provided under the 1990 Americans with Disabilities Act and 2008 Genetic Information Nondisclosure Act to allow genetic testing to be part of workplace wellness programs. What has privacy advocates concerned is that Obamacare allows employers to levy a hefty insurance surcharge on workers who don't participate in these ostensibly voluntary programs. (Fortune)
INDICATIONS
Biopharma cheers as Trump names Scott Gottlieb next FDA commissioner. Did you feel that gust? No, it's not the Snowmageddon slated to hit the East Coast tomorrow - it's the collective sigh of relief from drug makers at President Trump's choice to nominate Scott Gottlieb as FDA chief. Gottlieb, a physician and conservative health policy thinker, has widespread backing from the biopharma industry thanks to his experience and stated desire to cut back regulations which may delay the drug approval process. For the industry, he strikes the right balance on regulations. Unlike other potential FDA nominees like libertarian Peter Thiel associate Jim O'Neill, Gottlieb doesn't believe that drugs which have proven to be safe (but not necessarily effective) should win FDA green lights. He has, however, criticized the approval pathway as one that is bogged down with red tape. (TIME)
Mylan seems slated for the first approved cancer drug biosimilar in the U.S. Mylan and Roche have come to an agreement about the former firm's copycat of Roche arm Genentech's breast cancer blockbuster Herceptin, potentially clearing the way for the first approval of a cancer drug biosimilar in the U.S. As I've previously reported, Mylan unveiled some impressive clinical data at last year's ASCO cancer meeting for its treatment, trastuzumab, which mimics the $6.8 billion-in-annual-sales Herceptin but will be provided at a lower price if approved. As part of the new settlement, Mylan is withdrawing challenges to Genentech's Herceptin patents. The firm's biosimilar could be approved by the fall.
THE BIG PICTURE
Anthem backs parts of Trumpcare. After an outpouring of industry opposition to the GOP's American Health Care Act, including from the biggest lobbies representing doctors, hospitals, seniors, and even insurers, at least one voice has (sort of) come out in support of the Obamacare replacement legislation - or, at least, certain sections of it. Insurance giant Anthem's CEO Joseph Swedish said that the bill "addresses the challenges immediately facing the individual market and will ensure more affordable health plan choices for consumers in the short term" by rolling back Obamacare taxes on health insurance, temporarily extending the availability of subsidies that offset out-of-pocket costs, and providing tax credits for consumers who want to buy plans outside of Obamacare's marketplaces. The Congressional Budget Office's assessment of the AHCA could be released as soon as today. (Fortune)
REQUIRED READING
How to Deal With Persistent Social Anxiety, by Laura Entis
Intel to Buy Israeli Self-Driving Car Company Mobileye for $15 Billion, by Lucinda Shen
India's New Maternity Leave Policy Puts the U.S. to Absolute Shame, by Claire Zillman
You Should Observe National Napping Day, by Barb Darrow
Psst, Your Company Is Watching You, by Robert Hackett
| Produced by Sy Mukherjee | |
| @the_sy_guy | |
| sayak.mukherjee@fortune.com |
Find past coverage. Sign up for other Fortune newsletters.











