An FDA-approved contact lens for kids has helped make CooperVision, a division of CooperCompanies (ticker: COO), one to watch when it comes to eye care.
With MiSight, the only FDA-approved soft contact lens specifically designed to slow the progression of nearsightedness in children, CooperVision has produced a market-leading product. The daily disposable, which hit the market in early 2020, is helping to create a profound shift in vision care for nearsightedness, technically known as myopia. This condition presently affects more than 40% of people in the United States, up from about 25% in 1971, and its incidence is expected to rise significantly in the coming years.
MiSight’s value proposition is twofold: The contact lenses act like any other vision correction, though offer kids more freedom to play sports and do other activities than they might have with glasses. But the act of wearing them also helps to prevent their nearsightedness from worsening.
“We’re correcting the myopia that they do have and at the same time we’re slowing the progression,” says Michele Andrews, CooperVision’s VP of professional and government affairs.
Other treatments to slow myopia progression do exist, but they are off-label. MiSight is the only FDA-approved treatment for this application in children, and the studies that were needed to meet the agency’s requirements mean there is gold-standard evidence from long running clinical trials demonstrating the effectiveness of the product in kids.
“Ever since it came out I’ve been prescribing it,” says Dave Anderson, an Ohio optometrist who specializes in treating myopia. Anderson was an early enrollee in the CooperVision course that teaches eye-care professionals about the new product, and he estimates that being able to offer the treatment has made a substantial difference for his business, as well as for his patients. “I definitely do get more referrals.”
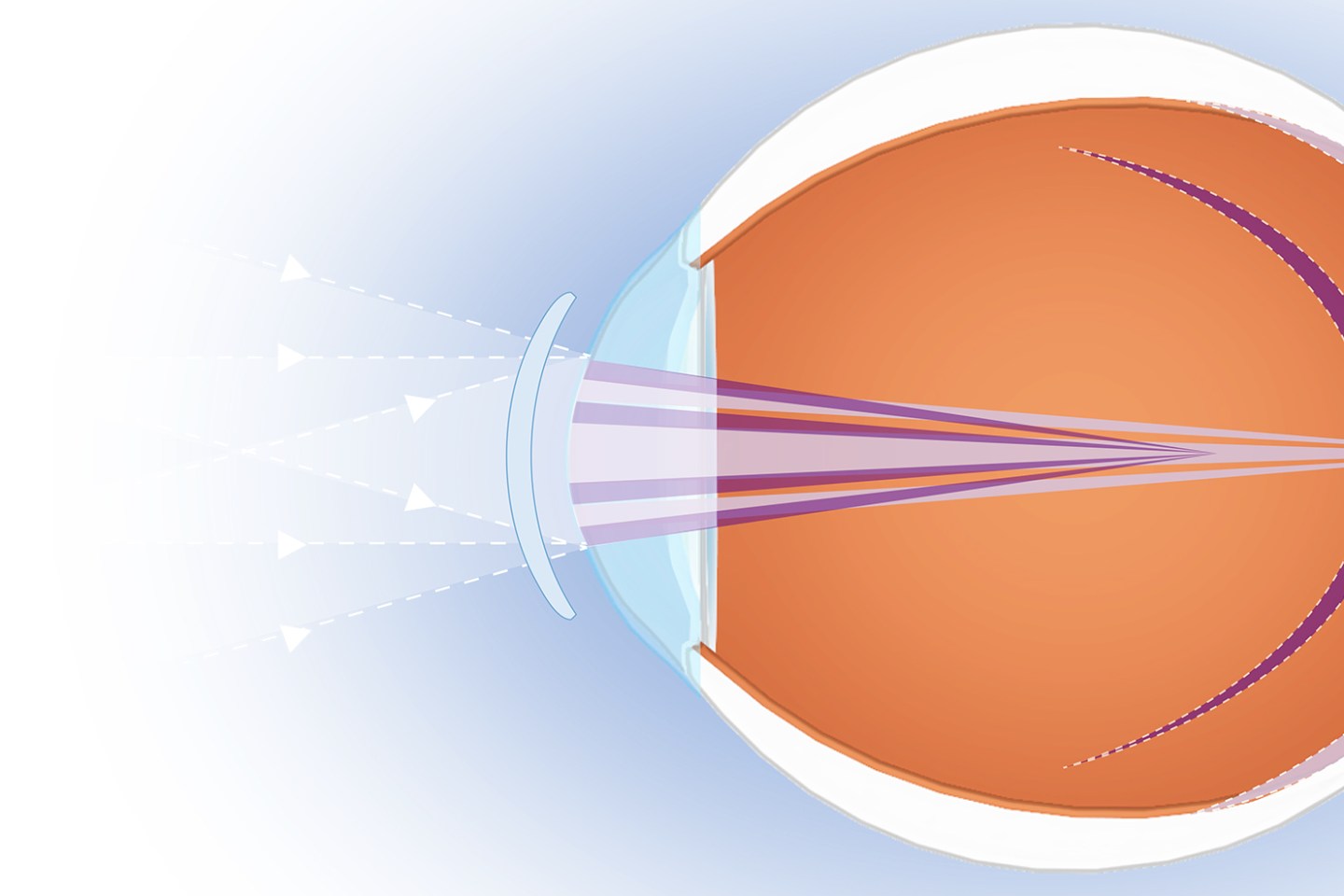
When someone develops myopia, their eye grows incorrectly, which leads the light entering the eyeball to refract incorrectly and converge in front of the retina instead of on it. That’s why things in the distance appear blurred.
In the past few decades, rates of nearsightedness have risen globally to epidemic proportions. Scientists still don’t know what exactly is behind this rise, although they believe a combination of genetics and environmental factors influence the condition. By 2050, estimates suggest that about half of the world’s population will be nearsighted—with nearly 10% being severely myopic.
In this context, myopia treatment is important not just for individual comfort and well-being, says Cheri Wiggs, an expert on low vision who oversees the National Eye Institute’s myopia program, but for public health as well.
“That’s particularly problematic not just because the myopic experience is more severe, but it’s also linked to a higher risk of developing eye conditions that lead to permanent visual impairment,” says Wiggs.
In other words: An epidemic of myopia could lead to an epidemic of retinal detachment, glaucoma, and the early development of cataracts. These issues already place a significant burden on global public health.
When it comes to myopia, Wiggs’s colleague Jimmy Le, who works on clinical research at the National Eye Institute, says he thinks about the three A’s: awareness, availability, and access. Many people around the world—and even in the United States—won’t be able to access MiSight, which comes at a much higher price point than traditional single-focus contact lenses or glasses.
Still, he says, “from NEI’s perspective, we’re always supportive of myopia studies.”
Although eyesight changes throughout a person’s life, the biggest changes usually happen between the elementary school years and late teens. Those are the years when many corrective lens wearers get their first pair, and they’re also the most important window for myopia control interventions. In fact, MiSight’s contact lenses are only FDA-approved if the treatment begins between age 8 and 12.
If treatment begins in that period, they’ve been shown to slow myopia progression by about 50%. The children who wore MiSight lenses in the study that led to FDA approval showed about half the increase in myopia of the control participants, who wore regular soft contacts. Their eyes showed 59% less progression away from ideal refraction and 52% less growth of eye length. The second condition is strongly associated with those dangerous eye problems later in life.
There’s no reason to think that they won’t still slow myopia progression if treatment begins later, says Jeffrey Walline, a professor of optometry at the Ohio State University, although the reduction may not be as significant.
Historically, optometrists weren’t able to do much more than monitor the progression of a patient’s myopia and prescribe stronger lenses as appropriate. But in the past decade or so, says Walline, research has significantly advanced strategies to actively slow the progression of nearsightedness in those crucial years.
Treatments for adults, like laser eye surgery, actually reshape the eye so light refracts correctly. Myopia control techniques, on the other hand, intervene early to keep the eye from growing incorrectly to begin with.
Before MiSight hit the market there were a number of options that worked, but none had been designed specifically for this application.
Low concentration eye drops of a pupil-dilating substance called atropine have been shown to be extremely effective for many patients when administered daily, Walline says. Sleeping in hard contact lenses that reshape the eye, called orthokeratology, is also effective, he says. CooperVision makes a lineup of orthokeratology lenses, including one specifically aimed at children.
These options have their drawbacks. Not all children are comfortable wearing the hard lenses, which also require a high level of cleanliness, says Walline. Some patients don’t respond to the atropine treatment, and it requires an additional daily commitment.
For those reasons, options that perform a dual function, like contact lenses or glasses, are preferred. Some other bifocal contact lenses, prescribed off-label, work about as well as the new CooperVision product, Walline’s research has found. Walline is one of the lead researchers on the NEI-funded BLINK study, which looks at how dual-focus contact lenses (in the case of the study, monthlies) can help slow the development of myopia.
But prior to the introduction of MiSight to the market, optometrists didn’t have a purpose-built option to offer parents who wanted to slow the progression of their child’s myopia.
The MiSight contact lenses were designed with children in mind and have been trialed internationally since 2009. They’re daily disposables, which removes many hygiene concerns, and the product is supported by an app for parents and optometrist visits once every three months to check on any prescription changes—as well as pricing that accommodates as many contact lens fittings as a child needs.
“I felt like it was a really good addition to my portfolio to help with myopia management,” Anderson says. The backing of the FDA and the published studies showing continued benefit are “very nice to have,” he adds.
That speaks to the value of the purpose-designed product, says Ashley Wallace Tucker, a Houston optometrist whose interview was arranged by CooperVision. “We all believe that daily disposable lenses are the best option for children,” she says.
The product was a big part of CooperVision’s solid year. A whopping 165% rise in sales in Q4 2021, together with 40% growth in its orthokeratology lines, the company’s other myopia management products, contributed to a strong finish from CooperCompanies. In 2022, the company expects this segment to grow to approximately $100 million, according to a report from Zacks Equity Research.
“When it comes to MiSight, the company has made excellent progress with its key accounts and has entered into new pilot programs with retailers and buying groups globally, thereby continuing to gather steam,” the report notes.
Wallace Tucker says she was “chomping at the bit” to start using MiSight for her young patients and had a waiting list of people who wanted to try it after other forms of myopia control didn’t work for their children.
She did her first fitting not long after the product came on the market, around May 2020. In the time since, Bellaire Family Eye Care, her practice, has fitted about 80 kids with the soft contact lenses. At $2,000 for a year of concierge-style care, including the lenses themselves and whatever assistance the patient needs, she says, “it’s been a huge practice builder for us.”
The idea of not only correcting myopia but controlling it is fairly new. Her patients’ parents, who are often myopic themselves, don’t generally know about it, says Wallace Tucker. Once she explains, she says, they’re often eager to explore options that will leave their kids with better eyesight.
But consumer awareness has been a heavy lift, says Andrews, the CooperVision VP. The company has been eager to educate eye care professionals about how to discuss myopia management and how to use MiSight specifically, she says. More than 5,000 U.S. eye doctors have gone through CooperVision’s orientation program, she says, and are now using MiSight for their patients.
In addition, the company launched a large direct-to-consumer campaign in summer 2020, bringing on celebrities like Sarah Michelle Gellar and child performers Alan Kim and Madison Reyes to act as spokespeople.
The market for myopia control products is far from saturated. Research has shown that different patients prefer different myopia control lenses, suggesting this market segment would benefit from diversification.
In addition, adults with myopia are experiencing worsening vision at higher rates than they used to, often heading toward the danger zone of high myopia. This has led some to prescribe MiSight off-label for their adult patients: Walline says this topic has come up a number of times when he talks about his work in professional settings.
“Myopia control strategies that are used for children may work for adults, but it’s not guaranteed until we have more data,” says the NEI’s Le.
“We are seeing certainly a greater attention to myopia and its progression because of the last couple years and how we’ve had to live,” says Andrews. She declined to share details of what CooperVision is working on for MiSight’s next act, “but we do have a very active pipeline about ideas around myopia progression and where we might go with MiSight,” she says.
CooperVision is also working on getting myopia control glasses to market in the United States. Last year, it partnered with France-based eyewear multinational EssilorLuxottica to buy SightGlass Vision, a company that makes myopia control eyeglasses, at present for the international market.
That’s the biggest need U.S. eye doctors have right now for myopia control, says Wallace Tucker. “We need a really good spectacle option,” she says.