The Food and Drug Administration (FDA) on Monday granted full approval for Pfizer’s COVID vaccine. The long anticipated move means that Pfizer/BioNTech’s shot is no longer just available under an FDA “emergency use authorization,” or EUA, a designation the coronavirus vaccine has carried since it became the first to be authorized by regulators on Dec. 11, 2020, but under a full official approval for adults 16 and older. (Americans ages 12 to 15 can still receive it through the existing FDA emergency authorization.) Government and public health officials hope full approval will spur the vaccine hesitant to finally get their shots, but what does this approval mean for businesses, and what impact will it really have on vaccinations?
Regulators and public leaders are banking on the approval to assuage Americans concerned about possible harm or side effects from a COVID vaccine, or those who are just generally mistrusting of the news media and the numerous studies establishing the vaccine’s safety and efficacy, and to persuade them to join the immunization campaign. The approval also has the federal government taking a more aggressive stance, with the Pentagon mandating all active duty personnel to be vaccinated hours after the approval’s announcement, and President Joe Biden urging business and government leaders to seize the moment and step up immunization requirements in their communities and workforces.
Pfizer’s COVID vaccine, as the first to reach the market, is the one that’s been administered most in America. According to the latest Centers for Disease Control (CDC) data, 56% of all vaccinated U.S. adults had at least one dose of Pfizer’s product (which will be marketed under the brand name Comirnaty). A full FDA approval is different from an emergency authorization since full approval requires more robust, long-term study data for establishing a vaccine’s safety and effectiveness. EUAs are granted in emergencies based on the “best available evidence” at the time with close monitoring of new information, but an FDA-approved treatment is one “the agency has determined, based on substantial evidence…is effective for its intended use, and that the benefits of the drug outweigh its risks when used according to the product’s approved labeling.” Moderna’s jab is in the earlier stages of the approval process, and both of the mRNA vaccines have shown high effectiveness in preventing COVID-related hospitalizations and deaths, even though the Delta variant may weaken the effectiveness of Pfizer’s vaccine when it comes to infections and COVID symptoms that don’t require hospitalization.
That could theoretically nudge people to give COVID shots the benefit of the doubt. “While millions of people have already safely received COVID-19 vaccines, we recognize that for some, the FDA approval of a vaccine may now instill additional confidence to get vaccinated,” said FDA acting commissioner Janet Woodcock in a statement Monday. Others such as the bipartisan duo of Arkansas Republican Gov. Asa Hutchinson and Democratic Gov. Phil Murphy of New Jersey, who are the respective chair and vice chair of the National Governors Association (NGA), shared the sentiment.
“We have heard from many of our residents as we have traveled across our states and territories, held town halls, and met with residents, who are hesitant to get vaccinated because they are awaiting full approval of the vaccines. FDA’s approval of the Pfizer vaccine is another tool that will help combat hesitancy by addressing some of the objections and safety concerns that have been expressed,” Hutchinson and Murphy said in a joint NGA statement. “We are hopeful that with today’s FDA announcement more individuals will be encouraged to get vaccinated as we continue to battle this pandemic.”
But whether or not American individuals and firms will embrace that spirit is the million-dollar question in the coming weeks. Biden on Monday made his stance clear to businesses and government officials to use the FDA’s Pfizer vaccine approval as an impetus to get their workforces and communities vaccinated. “I’m calling on more companies in the private sector to step up with vaccine requirements that will reach millions more people,” Biden said in statements from the White House on Monday. “If you’re a business leader, a nonprofit leader, a state or local leader who has been waiting for full FDA approval to require vaccinations, I call on you now to do that. Require it. It only makes sense to require a vaccine to stop the spread of COVID-19.
“If you’re one of the millions of Americans who said that they will not get the shot until it has full and final approval of FDA: It has now happened. The moment you’ve been waiting for is here,” the President added.
To that end there’s been a growing number of companies mandating COVID vaccines or requiring negative tests and other safety measures for many of their employees to return to shared office spaces as the coronavirus Delta variant continues to spread. Those include the likes of Microsoft, Tyson Foods, Uber, Walmart, Walgreens, United Airlines, and UPS.
Other firms, such as Delta Air Lines, that have previously said the lack of full FDA approval was holding them back from vaccine mandates have yet to publicly announce whether Monday’s decision has moved the needle on their policies—for both employees and customers. “It’s very difficult for us to come in and mandate a vaccine that isn’t even federally approved yet; the authorization hasn’t been final yet, so stay tuned,” Delta CEO Ed Bastian told CNBC in early August about the company’s decision to not mandate vaccines for customers on domestic flights.
Now businesses like Delta could very well move in that direction, and the federal government can use its muscle to put financial pressure on organizations as the Biden administration did when it threatened to cut off federal funding to nursing homes whose staff members aren’t vaccinated. Vaccine requirements for all federal employees are under consideration by the Biden administration. State and local governments that have yet to issue such mandates for staff or visitors at public schools, per CDC recommendations, could also follow suit after the approval, as New Jersey and New York City did on Monday. Surveys have indicated that most U.S. employers plan to require some if not all workers to be vaccinated—barring religious or medical objections—and polling over the past month has shown a shift in overall public support toward vaccine mandates, although workers remain divided on the issue.
There’s also the more fundamental question of how individual Americans will actually react to the FDA Pfizer approval. There’s some evidence that the Delta variant’s rise has persuaded some who initially said they wouldn’t get a COVID shot to change their minds, and that full FDA approval could influence others on the fence. But it’s a nuanced bit of public health psychology, and an approval may not even be the most effective persuasion method, as data from the Kaiser Family Foundation (KFF) think tank’s COVID tracker illustrates. While more than three in 10 surveyed adults (including nearly half of those who said they wanted to “wait and see” more about COVID vaccines before making a decision) recently said an FDA approval would make it more likely that they would get immunized, that figure could be misleading, according to KFF’s analysis.
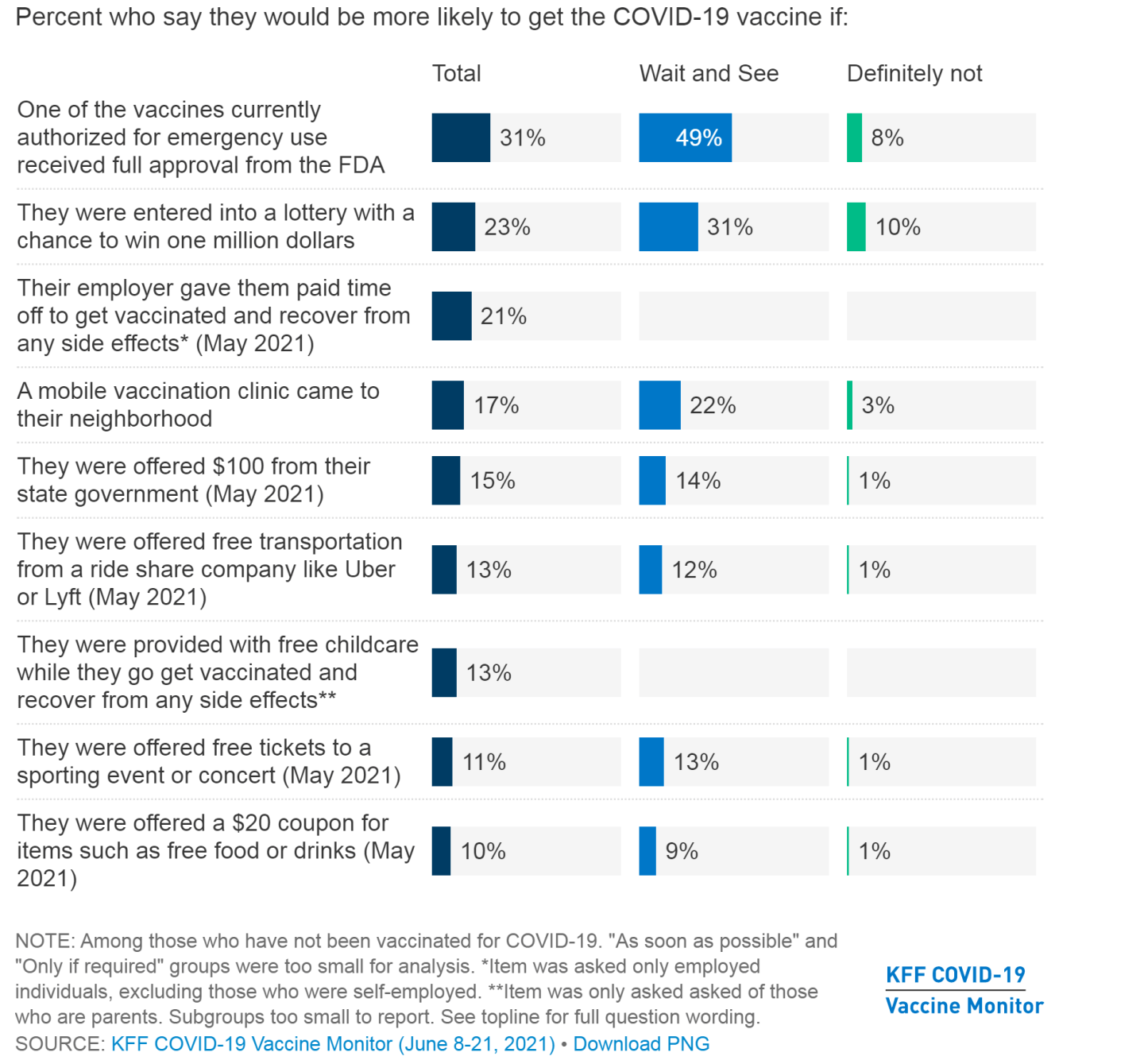
“This finding likely suggests that FDA approval is a proxy for general safety concerns, as two-thirds of adults (including a large majority of unvaccinated adults) either believe the vaccines currently available in the U.S. already have full approval from the FDA or are unsure whether they have full approval or are authorized for emergency use,” wrote the survey authors.
Nearly half of unvaccinated American adults said they would “definitely not” get a COVID vaccine this year in a KFF survey at the end of July. The coming months will reveal whether or not this key regulatory win for Pfizer softens their defenses and gives businesses the cover and confidence they need to get workers vaccinated, or whether a combination of factors—including the Delta variant crisis—is what’s necessary for more shots to reach arms. The good news is that vaccination rates have been on the rise since the end of July, when Delta’s impact began to seep into the public consciousness. The seven-day moving average of daily COVID vaccine doses administered was up to 731,854 per day as of Aug. 18, according to CDC data, up from 575,812 at the end of July.
Subscribe to Fortune Daily to get essential business stories straight to your inbox each morning.











