If you were to track the upgrades for your Apple iPhone or Toyota Prius from their introduction to today, you will see a familiar arc in the technology industry: performance multiplies, the product is refined, jobs are created, even entire industries are reworked.
Consider, for example, that the iPhone’s theoretical maximum download speed on cellular networks went from 1 megabyte per second for the 2007 “2G” iPhone to 300 mbps for today’s 5s model. Its display more than doubled in pixel density, its camera transformed from cheap afterthought to serious photography tool, and its software capabilities are far more robust than when the device was introduced. (Even the App Store is a second-generation feature.)
Similarly, Toyota’s Prius hybrid car evolved from a neighborhood oddity (and celebrity eco-accessory) in 2000 to a best-selling vehicle in Japan and California. The engine in today’s model is 20 percent lighter (and offers 20 percent more total horsepower) than the original. Its distance-per-charge is longer. Without the Prius, it can be argued, there would be no Tesla.
There’s is one component of all of these things that hasn’t changed in that time period: the lithium-ion battery. Whether in the iPhone, the Prius, and even the Tesla Model S, the Li-ion battery is essentially made of the same stuff as those first introduced by Sony in 1991. That’s not to say that innovation hasn’t happened around them, of course. Device-makers have become better at charging them, cooling them, and controlling how much power they draw into our phones, cars, laptops, and USB gadgets. But they’re still largely the same battery. Even Tesla’s $5 billion plans for a “giga”-sized battery factory involve the manufacture of—you guessed it—lithium-ion packs.
Upon further investigation, there is little consensus on what kind of battery technology may replace lithium ion. There aren’t even rumors.
To find out why, Fortune posed a simple question to five established researchers working on next-generation batteries, a behavioral economist, and a battery industry executive: Why is battery technology moving so much slower than hardware?
As you’ll soon find out, the answer is one part chemistry, one part psychology, and two parts the answer to a counter-question: Who really wants to be the first to drive with a new type of battery that hasn’t benefited from two decades of development?
Today’s battery tech: dense, hot, tricky
Lithium-ion battery technology is in many ways the workhorse of portable power.
Lithium’s atomic number is three, which, if you remember middle-school chemistry, means that it has three protons, is very lightweight, and can be packed more densely than any element other than hydrogen or helium. Lithium is a known quantity to chemists, says Carlo Segre, professor of physics at the Illinois Institute of Technology in Chicago, and we mostly understand how it flows inside a battery.
“I think it really boils down to, the reason lithium is so good, is that it’s very light, and you can get it through a membrane very easily,” Segre says. “And the potential difference (voltage) you can generate is one of the highest we know.”
It’s not just lithium that goes into a Li-ion battery. The element gets mixed with magnesium (for personal gadgets and vehicles), iron phosphate (for heavy-duty work), and other metals. That mixture flows into another material to create voltage: graphite, titanium solutions, silicon, and different forms of carbon, depending. In most non-industrial devices used in relatively safe conditions, you have lithium manganese oxide flowing into graphite, because those materials are cheap, relatively safe, and dense.
But there are quite a few problems with the old faithful. The process generates heat in a dense space, requiring some kind of cooling system. (A tremendous amount of work went into Tesla’s car-length liquid cooling rig, for example.) The electrolyte that conducts lithium’s flow adds weight. The cells lose their capacity over time. Charging the battery, which makes the lithium flow back, could be quicker. And though it’s rare, we have seen that tightly packed batteries full of fluids, made very hot, can sometimes puncture or explode.
What we might use next: air
Chandrasekhar “Spike” Narayan, director of science and technology at IBM Research, is part of the Battery 500 Project. The goal is to get batteries to power a car of average cost on a 500-mile trip. IBM (IBM) won’t build the batteries itself, but will partner with manufacturing and consumer companies to get them into the wild.
After years of work, Narayan sees a future for lithium-air technology, which replaces graphite and other metals with oxygen, refreshed by the car itself. Such batteries could be lighter, safer, and last far longer. But working with new mixtures, pushing them into new materials, and seeing how safe they are over thousands of charge cycles takes a very, very long time.
“There is no guiding principle that suggests you get improvement from year to year,” Narayan says. “There is no magic knob you can turn. The only way we can get to that kind of paradigm is a completely new kind of chemistry, and innovation doesn’t work like that.”
Currently, lithium-air batteries have to overcome problems with blockages, internal rust, and stability. Even if air batteries are smoothed into a viable product, Narayan sees a future where battery technology is no longer one-size-fits all. “It may not be a great technology for power grid storage, for example. Especially when there is a size requirement, we may see differentiation among battery types soon.”
What we can do in the meantime: get cheaper
Kevin Bai and Xuan “Joe” Zhou at Kettering University work in labs and in battery industry research, but they talk like car shoppers than laboratory wonks. With the hybrid vehicles of today, Zhou notes, there are lots of trade-offs, in several ways.
“Right now [hybrid] batteries are selling for $500 to $600 per kilowatt hour, but they should be $200,” Zhou says. “And every dollar you spend in the battery is another dollar in cooling. If the car needs a $6,000 battery, it’s a $6,000 cooling system.” What’s more, Bai notes, the size of such a battery eats up trunk or seating space. The scientists agree that an electric vehicle should feel like less of a financial albatross.
But it’s anybody’s guess as to which current materials may work out to have the safest, coolest, and most lightweight mix, while still selling for less than today’s offerings.
Zinc-air batteries, used in hearing aids today, are seeing renewed interest, especially given zinc’s easy availability. The same goes for sodium-air, which are cheaper and easier to assemble, if not as potentially powerful as lithium-air. There are also attempts to replace the graphite and carbon solids in batteries with silicon, though silicon isn’t cheap. Or we might just improve the cost and performance of the lithium-iron batteries in our drills and motorcycles in the meantime.
In many ways, Bai says, building larger battery plants, better battery management tools, and a smarter power grid for charging is going to bear greater fruit than waiting on one or another chemical combo to pay off.
“We are actually very far away from a brand-new battery for vehicles,” Bai says. “The automotive industry, they must feel they can stand behind 10 years of testing before they are comfortable trying a new material.” It will be at least 2020, he says, before you see zinc-air batteries in the first four-wheeled vehicles–and then a long while more before that battery technology matures.
What we can do in the future: nano-engineer materials
Don’t give up on lithium-ion just yet, says Partha Mukherjee, a professor at Texas A&M University and leader in the American Society of Mechanical Engineers’ Nanoengineering for Energy and Sustainability group. We might still be using it, but with materials that have gained some new powers in the lab.
Nanoengineers might dig into the molecular structure of battery materials to speed up how they transfer more voltage per cell. There might be a change in the way the electrolyte conveys lithium ions so that “traffic jams” don’t occur and charging is much faster. You could design a thinner, stronger, but still flexible membrane for batteries that allows for swelling under heat but never breaks. Or go for broke and develop a material that absorbs more lithium ions than carbon, air, or any material we know.
“The fundamental question we need to ask is, ‘How about starting from the bottom up?” Mukherjee says. “That is the mesoscale paradigm that must be addressed. Can we make materials that are more tolerant of what we need batteries to do?”
In the meantime: get perspective
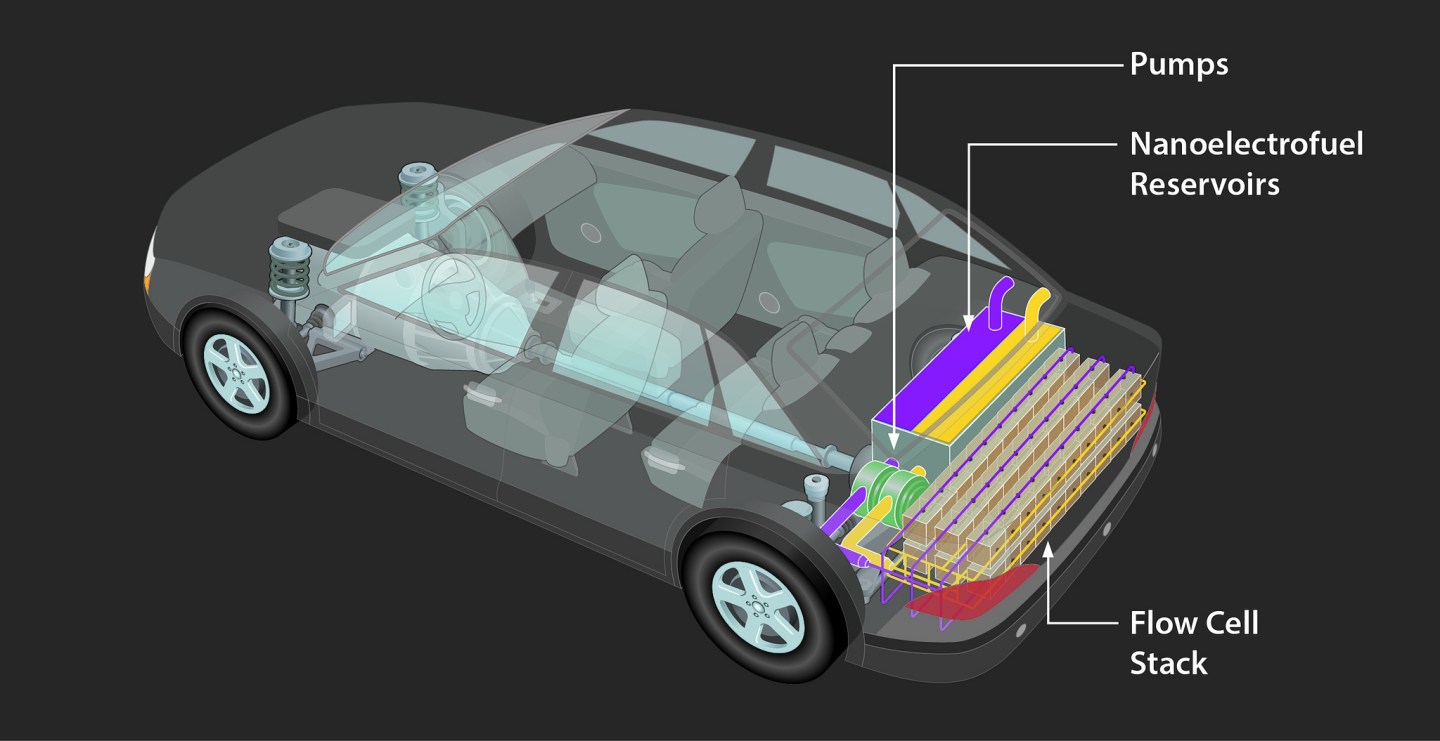
A year ago Segre, of the Illinois Institute of Technology, received a $3.4 million prize from the U.S. Department of Energy to develop a “flow battery” for car applications. Flow batteries store their active chemicals in external tanks and pass it through the battery structure itself. Segre’s work focuses on developing a liquid that is reactive and powerful enough to compensate for the liquid weight trade-off.
A flow battery might work in cars and power grid applications, but it will never work for a phone or laptop. Segre, like most researchers, knows it will be a long series of experiments until researchers hit upon a few different material combinations for batteries. In the meantime, “It’s especially frustrating for most of us because the battery dies, the capacity drops, after a couple years, while the electronics it powers could go on and on.”
For decades, we lived within Moore’s Law, which predicted that the number of transistors packed into a processor would double every two years, providing a steady gallop of technology improvement. We are now approaching a point at which transistors are near atomic-scale, chips can’t fit many more processors, and we’re unhappy with having the same kinds of batteries in our devices.
In other words, when it comes to physics, there’s no app for that. Which can be a bitter pill for tech-savvy consumers to swallow as they become acclimated to regular advancements in every other part of their electronic devices, says Michal Ann Strahilevitz, a professor of marketing at Golden Gate University.
“Adapting to upgrades is easy, and the more you are upgraded, the more you expect further upgrades,” Strahilevitz says. “In a world where [gadgets] keep getting better and more efficient, we feel we have a right to that. We ask, ‘Why can’t they be more wonderful than this?'”